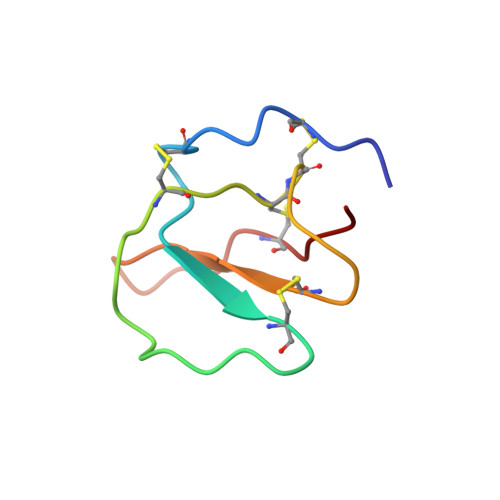
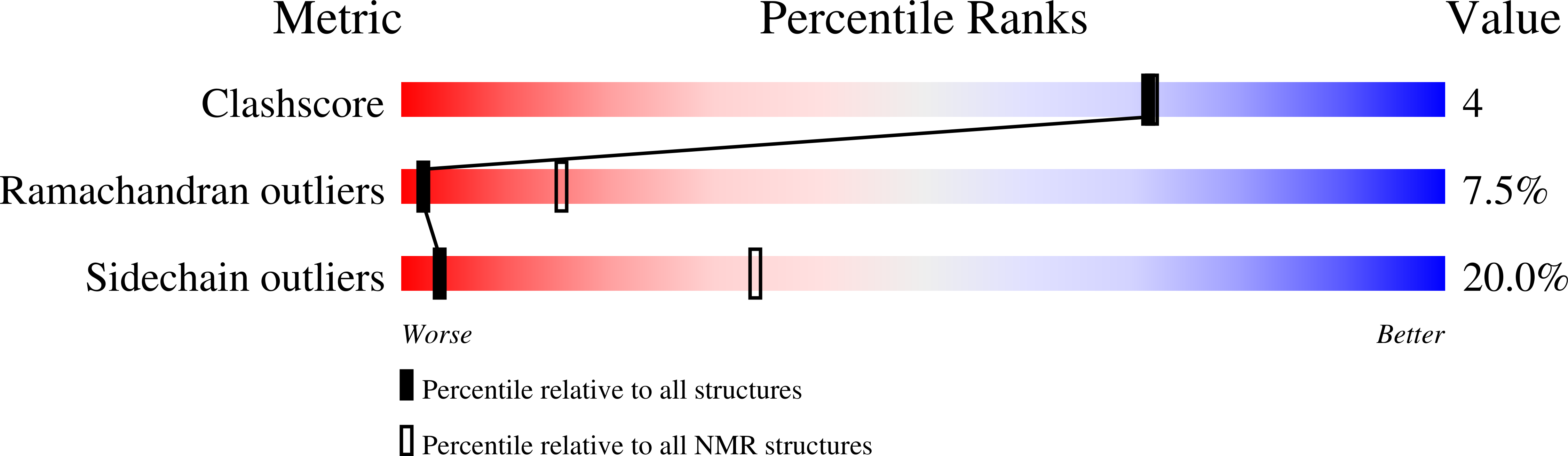Structure of a putative ancestral protein encoded by a single sequence repeat from a multidomain proteinase inhibitor gene from Nicotiana alata.
Scanlon, M.J., Lee, M.C., Anderson, M.A., Craik, D.J.(1999) Structure 7: 793-802
- PubMed: 10425681
- DOI: https://doi.org/10.1016/s0969-2126(99)80103-8
- Primary Citation of Related Structures:
1CE3 - PubMed Abstract:
The ornamental tobacco Nicotiana alata produces a series of proteinase inhibitors (PIs) that are derived from a 43 kDa precursor protein, NaProPI. NaProPI contains six highly homologous repeats that fold to generate six separate structural domains, each corresponding to one of the native PIs. An unusual feature of NaProPI is that the structural domains lie across adjacent repeats and that the sixth PI domain is generated from fragments of the first and sixth repeats. Although the homology of the repeats suggests that they may have arisen from gene duplication, the observed folding does not appear to support this. This study of the solution structure of a single NaProPI repeat (aPI1) forms a basis for unravelling the mechanism by which this protein may have evolved.
Organizational Affiliation:
Centre for Drug Design and Development, University of Queensland, St Lucia, Australia.