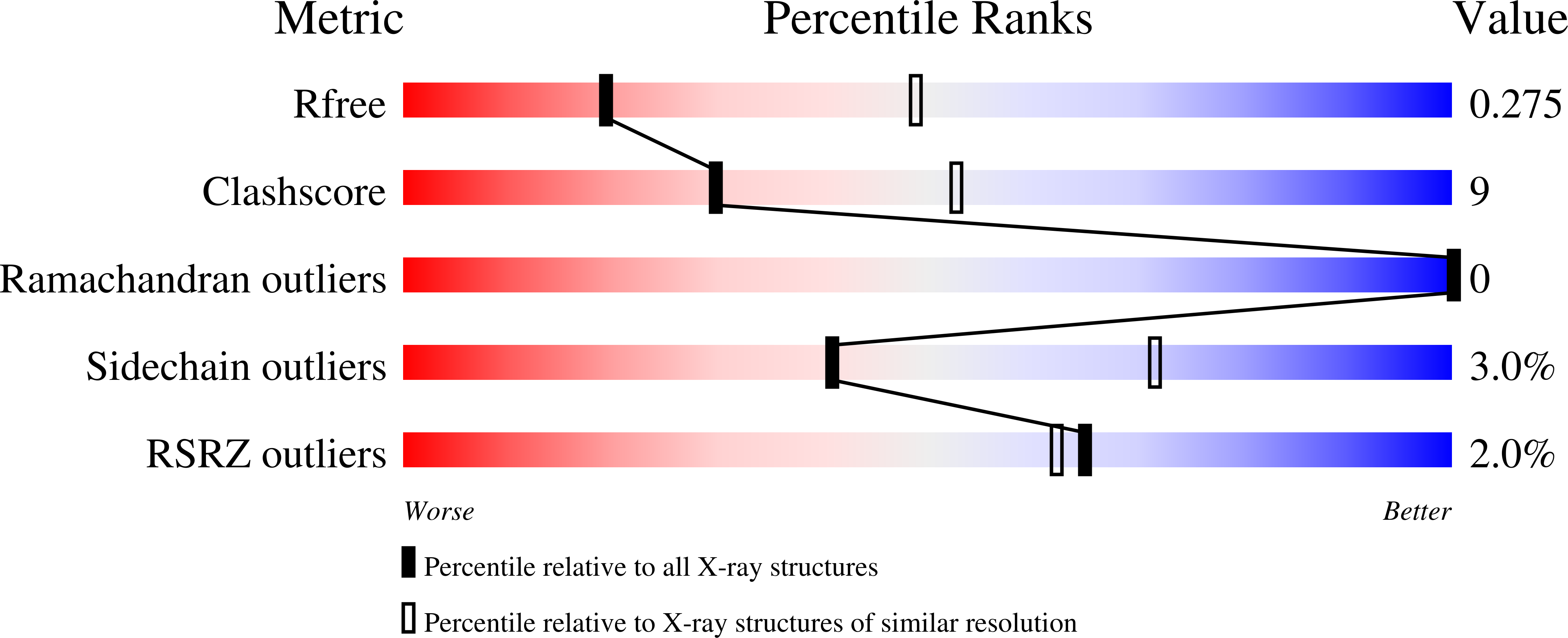
Effective mismatch repair depends on timely control of PCNA retention on DNA by the Elg1 complex.
Paul Solomon Devakumar, L.J., Gaubitz, C., Lundblad, V., Kelch, B.A., Kubota, T.(2019) Nucleic Acids Res 47: 6826-6841
- PubMed: 31114918
- DOI: https://doi.org/10.1093/nar/gkz441
- Primary Citation of Related Structures:
6D0Q, 6D0R - PubMed Abstract:
Proliferating cell nuclear antigen (PCNA) is a sliding clamp that acts as a central co-ordinator for mismatch repair (MMR) as well as DNA replication. Loss of Elg1, the major subunit of the PCNA unloader complex, causes over-accumulation of PCNA on DNA and also increases mutation rate, but it has been unclear if the two effects are linked. Here we show that timely removal of PCNA from DNA by the Elg1 complex is important to prevent mutations. Although premature unloading of PCNA generally increases mutation rate, the mutator phenotype of elg1Δ is attenuated by PCNA mutants PCNA-R14E and PCNA-D150E that spontaneously fall off DNA. In contrast, the elg1Δ mutator phenotype is exacerbated by PCNA mutants that accumulate on DNA due to enhanced electrostatic PCNA-DNA interactions. Epistasis analysis suggests that PCNA over-accumulation on DNA interferes with both MMR and MMR-independent process(es). In elg1Δ, over-retained PCNA hyper-recruits the Msh2-Msh6 mismatch recognition complex through its PCNA-interacting peptide motif, causing accumulation of MMR intermediates. Our results suggest that PCNA retention controlled by the Elg1 complex is critical for efficient MMR: PCNA needs to be on DNA long enough to enable MMR, but if it is retained too long it interferes with downstream repair steps.
Organizational Affiliation:
Institute of Medical Sciences, School of Medicine, Medical Sciences & Nutrition, University of Aberdeen, Foresterhill, Aberdeen AB25 2ZD, Scotland, UK.