The Antiviral Mechanism of an Influenza A Virus Nucleoprotein-Specific Single-Domain Antibody Fragment.
Hanke, L., Knockenhauer, K.E., Brewer, R.C., van Diest, E., Schmidt, F.I., Schwartz, T.U., Ploegh, H.L.(2016) mBio 7
- PubMed: 27965447
- DOI: https://doi.org/10.1128/mBio.01569-16
- Primary Citation of Related Structures:
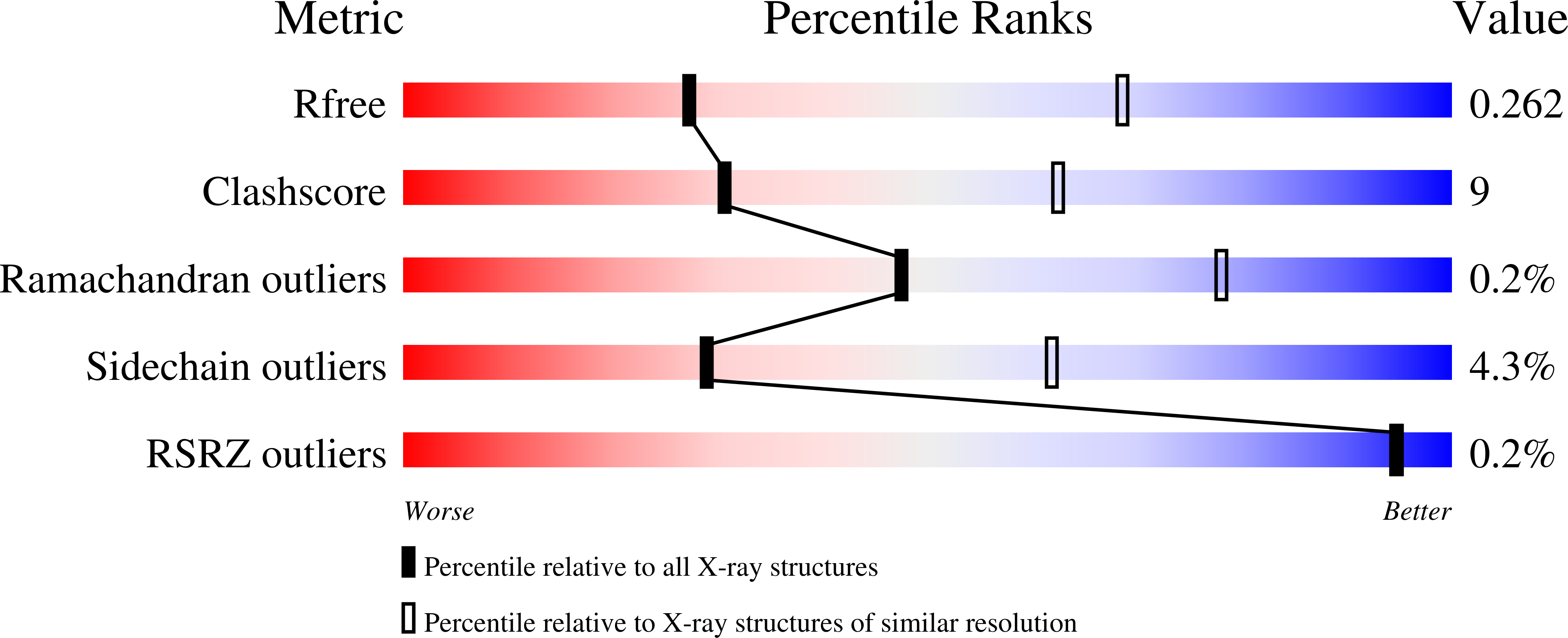
5TJW - PubMed Abstract:
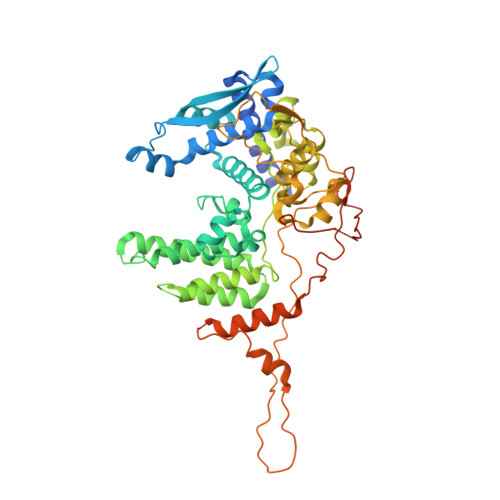
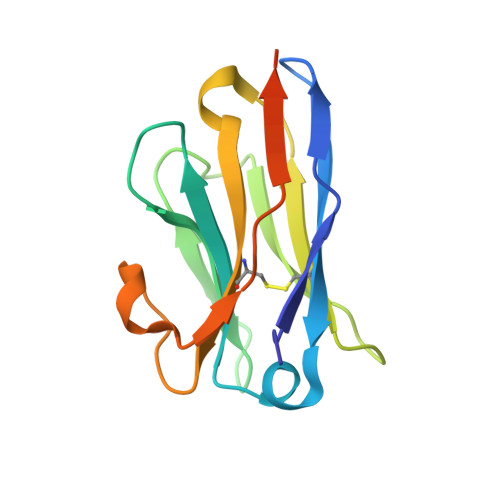
Alpaca-derived single-domain antibody fragments (VHHs) that target the influenza A virus nucleoprotein (NP) can protect cells from infection when expressed in the cytosol. We found that one such VHH, αNP-VHH1, exhibits antiviral activity similar to that of Mx proteins by blocking nuclear import of incoming viral ribonucleoproteins (vRNPs) and viral transcription and replication in the nucleus. We determined a 3.2-Å crystal structure of αNP-VHH1 in complex with influenza A virus NP. The VHH binds to a nonconserved region on the body domain of NP, which has been associated with binding to host factors and serves as a determinant of host range. Several of the NP/VHH interface residues determine sensitivity of NP to antiviral Mx GTPases. The structure of the NP/αNP-VHH1 complex affords a plausible explanation for the inhibitory properties of the VHH and suggests a rationale for the antiviral properties of Mx proteins. Such knowledge can be leveraged for much-needed novel antiviral strategies.
Organizational Affiliation:
Whitehead Institute for Biomedical Research, Cambridge, Massachusetts, USA.