Myristoylation drives dimerization of matrix protein from mouse mammary tumor virus.
Dolezal, M., Zabransky, A., Dostal, J., Vanek, O., Brynda, J., Lepsik, M., Hadravova, R., Pichova, I.(2016) Retrovirology 13: 2-2
- PubMed: 26728401
- DOI: https://doi.org/10.1186/s12977-015-0235-8
- Primary Citation of Related Structures:
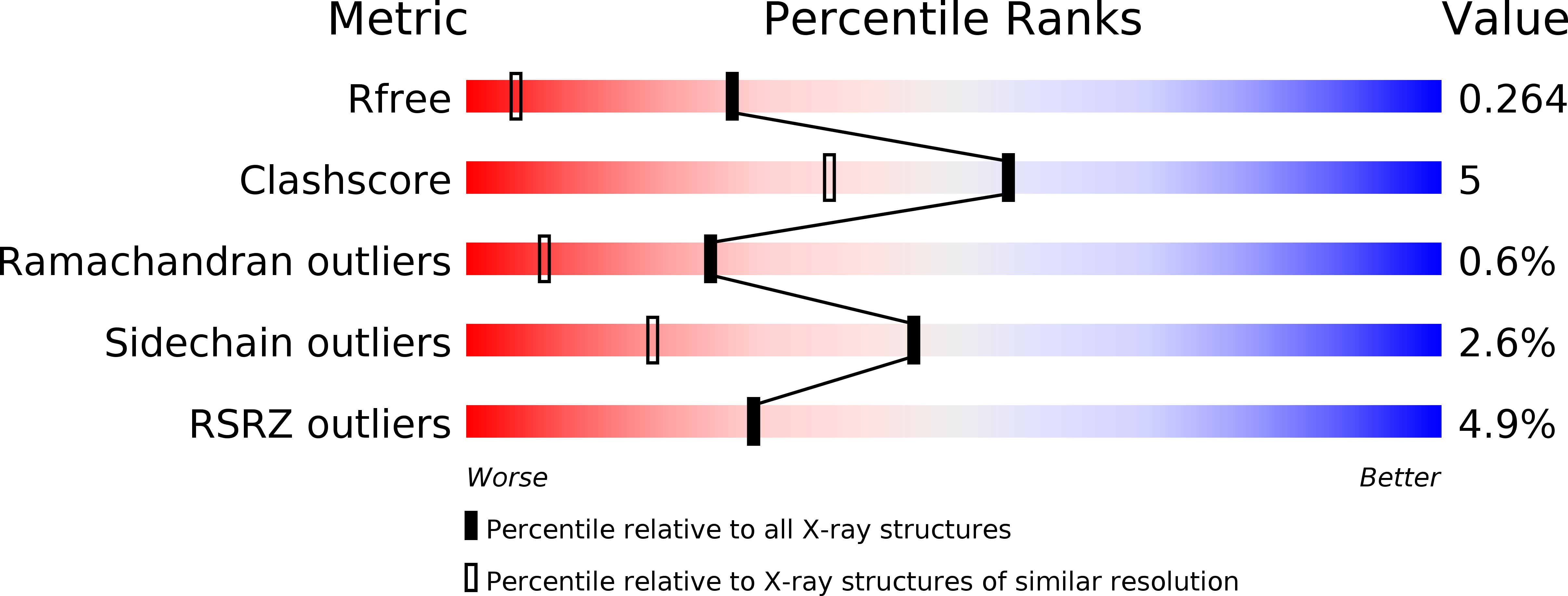
4ZV5 - PubMed Abstract:
Myristoylation of the matrix (MA) domain mediates the transport and binding of Gag polyproteins to the plasma membrane (PM) and is required for the assembly of most retroviruses. In betaretroviruses, which assemble immature particles in the cytoplasm, myristoylation is dispensable for assembly but is crucial for particle transport to the PM. Oligomerization of HIV-1 MA stimulates the transition of the myristoyl group from a sequestered to an exposed conformation, which is more accessible for membrane binding. However, for other retroviruses, the effect of MA oligomerization on myristoyl group exposure has not been thoroughly investigated.
Organizational Affiliation:
Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic, v.v.i., Flemingovo nám. 2, 166 10, Prague, Czech Republic. dolezal@uochb.cas.cz.