Mycofactocin-associated mycobacterial dehydrogenases with non-exchangeable NAD cofactors.
Haft, D.H., Pierce, P.G., Mayclin, S.J., Sullivan, A., Gardberg, A.S., Abendroth, J., Begley, D.W., Phan, I.Q., Staker, B.L., Myler, P.J., Marathias, V.M., Lorimer, D.D., Edwards, T.E.(2017) Sci Rep 7: 41074-41074
- PubMed: 28120876
- DOI: https://doi.org/10.1038/srep41074
- Primary Citation of Related Structures:
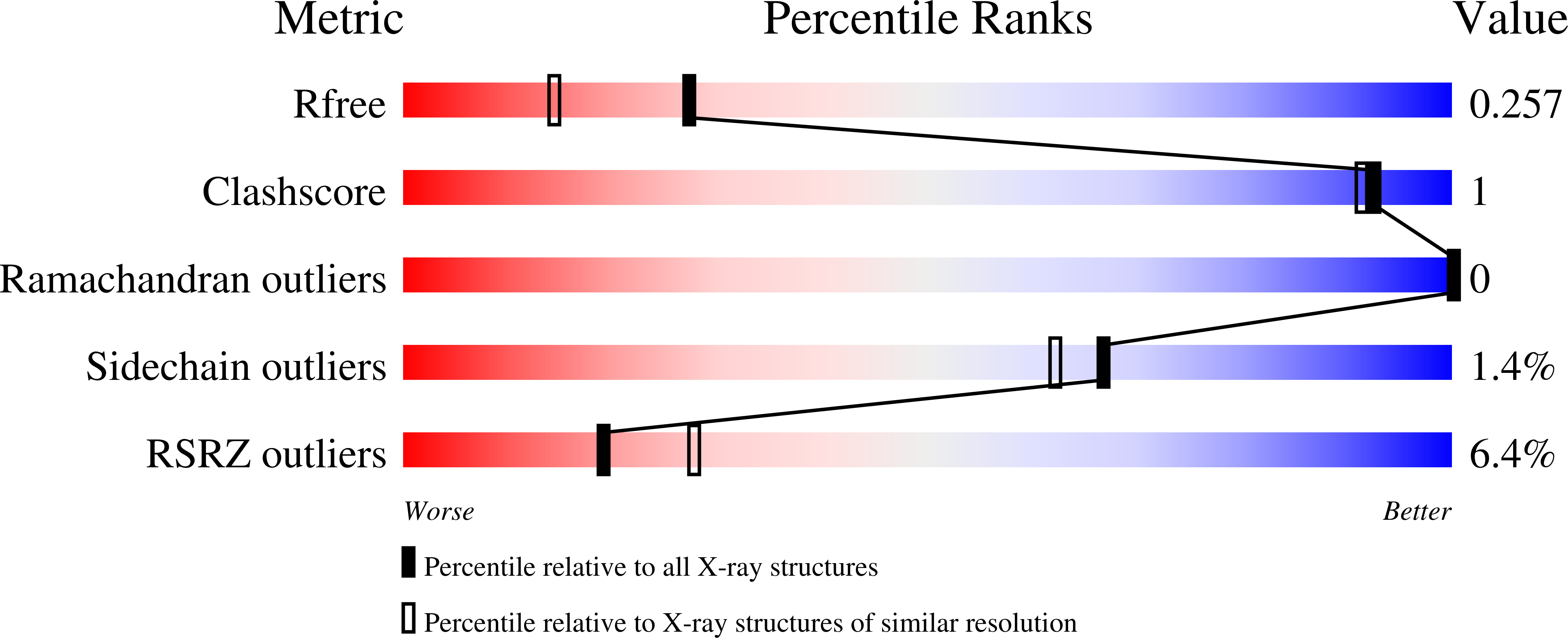
3OEC, 3PGX, 3PXX, 3S55, 3SX2, 3T7C, 3TSC, 4RGB, 5EJ2 - PubMed Abstract:
During human infection, Mycobacterium tuberculosis (Mtb) survives the normally bacteriocidal phagosome of macrophages. Mtb and related species may be able to combat this harsh acidic environment which contains reactive oxygen species due to the mycobacterial genomes encoding a large number of dehydrogenases. Typically, dehydrogenase cofactor binding sites are open to solvent, which allows NAD/NADH exchange to support multiple turnover. Interestingly, mycobacterial short chain dehydrogenases/reductases (SDRs) within family TIGR03971 contain an insertion at the NAD binding site. Here we present crystal structures of 9 mycobacterial SDRs in which the insertion buries the NAD cofactor except for a small portion of the nicotinamide ring. Line broadening and STD-NMR experiments did not show NAD or NADH exchange on the NMR timescale. STD-NMR demonstrated binding of the potential substrate carveol, the potential product carvone, the inhibitor tricyclazol, and an external redox partner 2,6-dichloroindophenol (DCIP). Therefore, these SDRs appear to contain a non-exchangeable NAD cofactor and may rely on an external redox partner, rather than cofactor exchange, for multiple turnover. Incidentally, these genes always appear in conjunction with the mftA gene, which encodes the short peptide MftA, and with other genes proposed to convert MftA into the external redox partner mycofactocin.
Organizational Affiliation:
National Institutes of Health (NIH), Bethesda, MD 20892, USA.