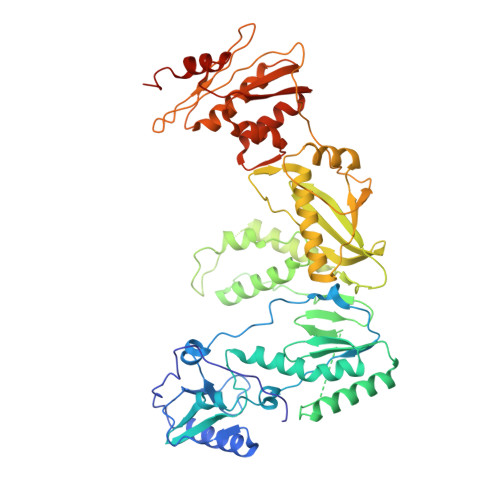
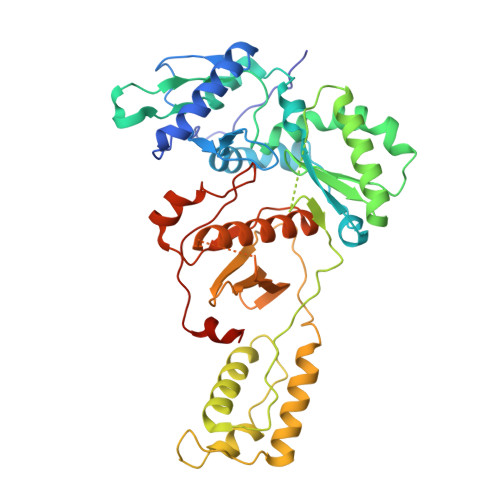
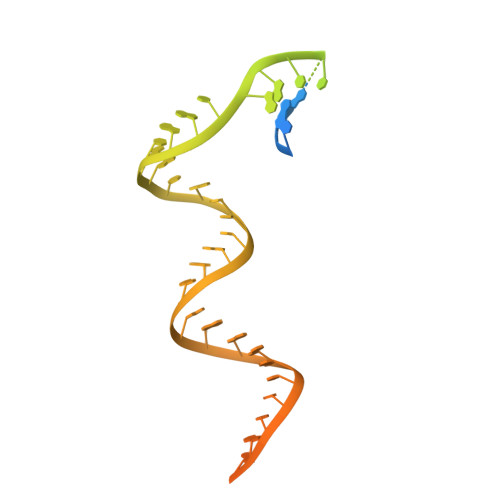
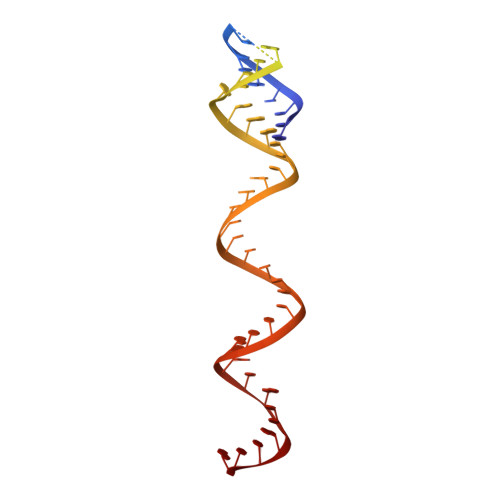
Distinct Conformational States Underlie Pausing during Initiation of HIV-1 Reverse Transcription.
Larsen, K.P., Choi, J., Jackson, L.N., Kappel, K., Zhang, J., Ha, B., Chen, D.H., Puglisi, E.V.(2020) J Mol Biol 432: 4499-4522
- PubMed: 32512005
- DOI: https://doi.org/10.1016/j.jmb.2020.06.003
- Primary Citation of Related Structures:
6WAZ, 6WB0, 6WB1, 6WB2 - PubMed Abstract:
A hallmark of the initiation step of HIV-1 reverse transcription, in which viral RNA genome is converted into double-stranded DNA, is that it is slow and non-processive. Biochemical studies have identified specific sites along the viral RNA genomic template in which reverse transcriptase (RT) stalls. These stalling points, which occur after the addition of three and five template dNTPs, may serve as checkpoints to regulate the precise timing of HIV-1 reverse transcription following viral entry. Structural studies of reverse transcriptase initiation complexes (RTICs) have revealed unique conformations that may explain the slow rate of incorporation; however, questions remain about the temporal evolution of the complex and features that contribute to strong pausing during initiation. Here we present cryo-electron microscopy and single-molecule characterization of an RTIC after three rounds of dNTP incorporation (+3), the first major pausing point during reverse transcription initiation. Cryo-electron microscopy structures of a +3 extended RTIC reveal conformational heterogeneity within the RTIC core. Three distinct conformations were identified, two of which adopt unique, likely off-pathway, intermediates in the canonical polymerization cycle. Single-molecule Förster resonance energy transfer experiments confirm that the +3 RTIC is more structurally dynamic than earlier-stage RTICs. These alternative conformations were selectively disrupted through structure-guided point mutations to shift single-molecule Förster resonance energy transfer populations back toward the on-pathway conformation. Our results support the hypothesis that conformational heterogeneity within the HIV-1 RTIC during pausing serves as an additional means of regulating HIV-1 replication.
Organizational Affiliation:
Department of Structural Biology, Stanford University School of Medicine, Stanford, CA 94305, USA; Program in Biophysics, Stanford University, Stanford, CA 94305, USA.