5VVX
Structural Investigations of the Substrate Specificity of Human O-GlcNAcase
- PDB DOI: https://doi.org/10.2210/pdb5VVX/pdb
- Classification: HYDROLASE
- Organism(s): Homo sapiens
- Expression System: Escherichia coli
- Mutation(s): Yes
- Deposited: 2017-05-21 Released: 2017-09-27
- Funding Organization(s): UW-Madison
Experimental Data Snapshot
- Method: X-RAY DIFFRACTION
- Resolution: 2.90 Å
- R-Value Free: 0.282
- R-Value Work: 0.200
- R-Value Observed: 0.204
This is version 1.3 of the entry. See complete history.
Macromolecules
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein O-GlcNAcase | A, B [auth C] | 504 | Homo sapiens | Mutation(s): 1 Gene Names: MGEA5, HEXC, KIAA0679, MEA5 EC: 3.2.1.169 (PDB Primary Data), 3.2.1 (PDB Primary Data) | 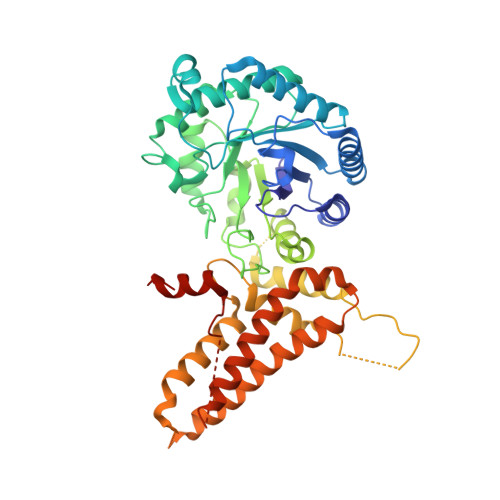 |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for O60502 (Homo sapiens) Explore O60502 Go to UniProtKB: O60502 | |||||
PHAROS: O60502 GTEx: ENSG00000198408 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O60502 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by: Sequence | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Lamin B1 | C [auth B], D | 13 | Homo sapiens | Mutation(s): 0 | 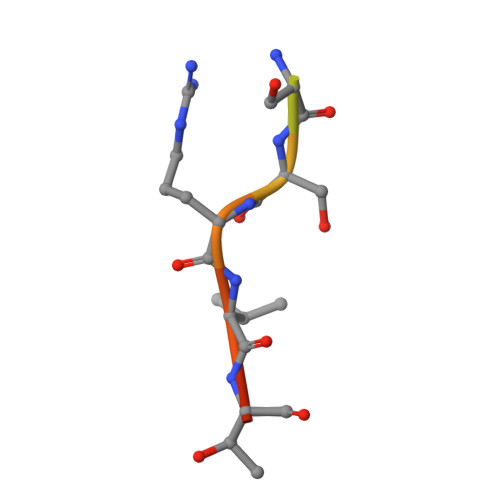 |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P20700 (Homo sapiens) Explore P20700 Go to UniProtKB: P20700 | |||||
PHAROS: P20700 GTEx: ENSG00000113368 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P20700 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Small Molecules
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Query on NAG | E [auth B], F [auth D] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
Experimental Data & Validation
Experimental Data
- Method: X-RAY DIFFRACTION
- Resolution: 2.90 Å
- R-Value Free: 0.282
- R-Value Work: 0.200
- R-Value Observed: 0.204
- Space Group: P 1 21 1
Unit Cell:
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 82.922 | α = 90 |
| b = 96.222 | β = 114.56 |
| c = 89.653 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |
Entry History & Funding Information
Deposition Data
| Funding Organization | Location | Grant Number |
|---|---|---|
| UW-Madison | United States | -- |
Revision History (Full details and data files)
- Version 1.0: 2017-09-27
Type: Initial release - Version 1.1: 2017-10-04
Changes: Database references - Version 1.2: 2020-07-29
Type: Remediation
Reason: Carbohydrate remediation
Changes: Data collection, Derived calculations, Structure summary - Version 1.3: 2023-10-04
Changes: Data collection, Database references, Refinement description, Structure summary













