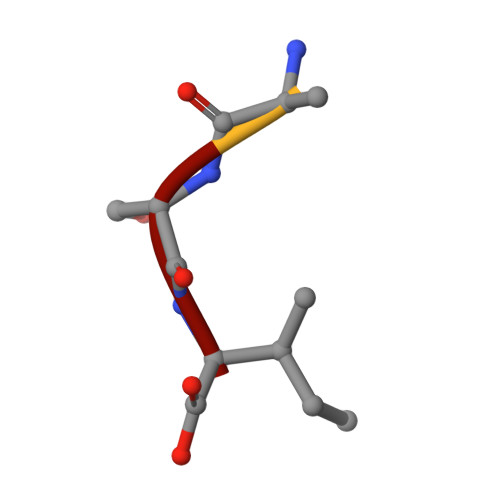
Molecular basis of ligand recognition by OASS from E. histolytica: insights from structural and molecular dynamics simulation studies
Raj, I., Mazumder, M., Gourinath, S.(2013) Biochim Biophys Acta 1830: 4573-4583
- PubMed: 23747298
- DOI: https://doi.org/10.1016/j.bbagen.2013.05.041
- Primary Citation of Related Structures:
4IL5, 4JBL, 4JBN - PubMed Abstract:
O-acetyl serine sulfhydrylase (OASS) is a pyridoxal phosphate (PLP) dependent enzyme catalyzing the last step of the cysteine biosynthetic pathway. Here we analyze and investigate the factors responsible for recognition and different conformational changes accompanying the binding of various ligands to OASS.
Organizational Affiliation:
School of Life Sciences, Jawaharlal Nehru University, New Delhi, India.