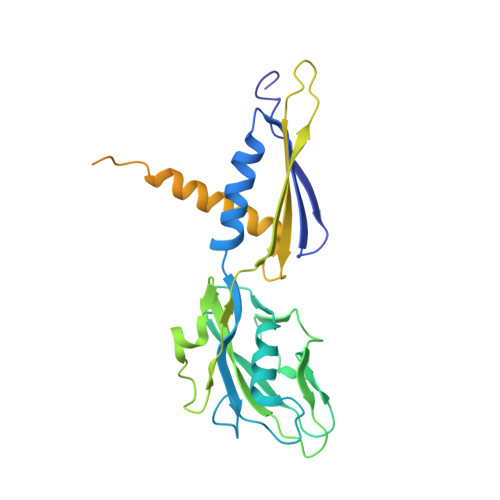
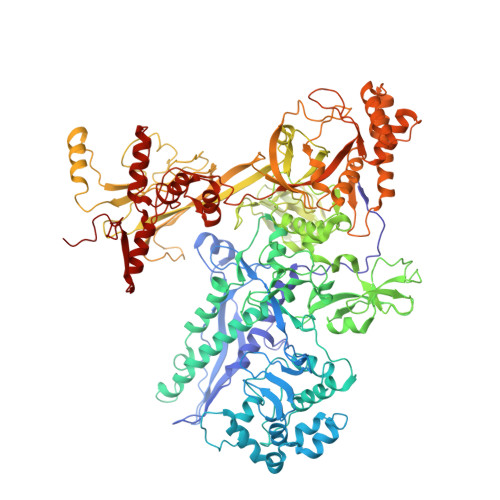
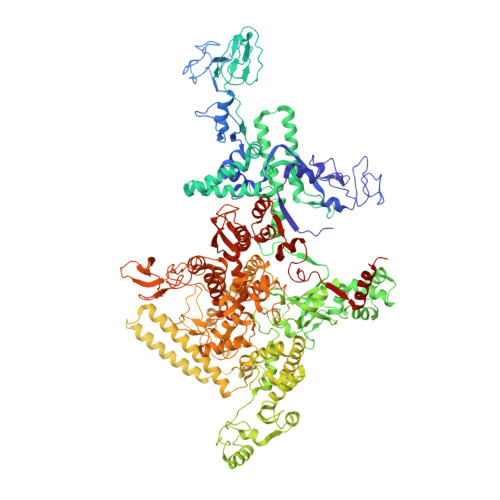
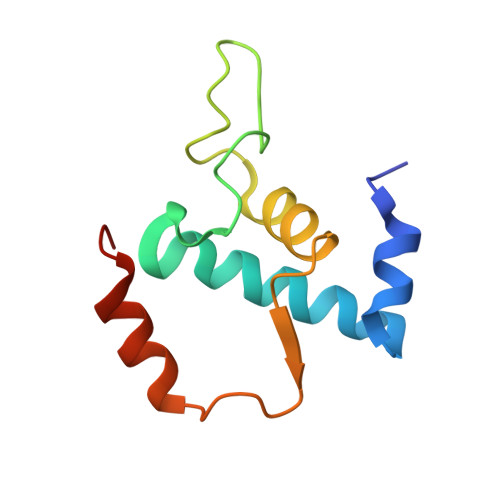
Transcription inactivation through local refolding of the RNA polymerase structure.
Belogurov, G.A., Vassylyeva, M.N., Sevostyanova, A., Appleman, J.R., Xiang, A.X., Lira, R., Webber, S.E., Klyuyev, S., Nudler, E., Artsimovitch, I., Vassylyev, D.G.(2009) Nature 457: 332-335
- PubMed: 18946472
- DOI: https://doi.org/10.1038/nature07510
- Primary Citation of Related Structures:
3EQL - PubMed Abstract:
Structural studies of antibiotics not only provide a shortcut to medicine allowing for rational structure-based drug design, but may also capture snapshots of dynamic intermediates that become 'frozen' after inhibitor binding. Myxopyronin inhibits bacterial RNA polymerase (RNAP) by an unknown mechanism. Here we report the structure of dMyx--a desmethyl derivative of myxopyronin B--complexed with a Thermus thermophilus RNAP holoenzyme. The antibiotic binds to a pocket deep inside the RNAP clamp head domain, which interacts with the DNA template in the transcription bubble. Notably, binding of dMyx stabilizes refolding of the beta'-subunit switch-2 segment, resulting in a configuration that might indirectly compromise binding to, or directly clash with, the melted template DNA strand. Consistently, footprinting data show that the antibiotic binding does not prevent nucleation of the promoter DNA melting but instead blocks its propagation towards the active site. Myxopyronins are thus, to our knowledge, a first structurally characterized class of antibiotics that target formation of the pre-catalytic transcription initiation complex-the decisive step in gene expression control. Notably, mutations designed in switch-2 mimic the dMyx effects on promoter complexes in the absence of antibiotic. Overall, our results indicate a plausible mechanism of the dMyx action and a stepwise pathway of open complex formation in which core enzyme mediates the final stage of DNA melting near the transcription start site, and that switch-2 might act as a molecular checkpoint for DNA loading in response to regulatory signals or antibiotics. The universally conserved switch-2 may have the same role in all multisubunit RNAPs.
Organizational Affiliation:
Department of Microbiology, The Ohio State University, 484 West 12th Avenue, Columbus, Ohio 43210, USA.