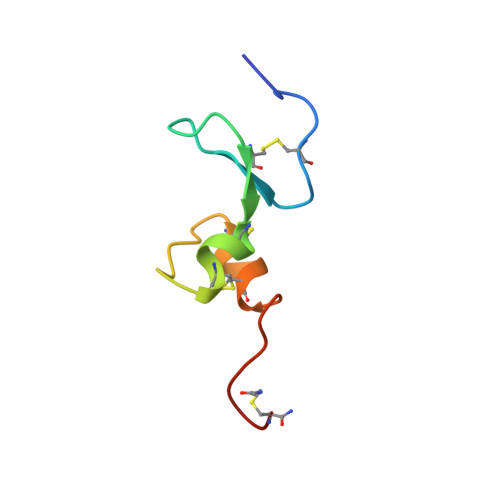
Structure of the extracellular domains of human and Xenopus Fn14: implications in the evolution of TWEAK and Fn14 interactions.
Pellegrini, M., Willen, L., Perroud, M., Krushinskie, D., Strauch, K., Cuervo, H., Day, E.S., Schneider, P., Zheng, T.S.(2013) FEBS J 280: 1818-1829
- PubMed: 23438059
- DOI: https://doi.org/10.1111/febs.12206
- Primary Citation of Related Structures:
2KMZ, 2KN0, 2KN1 - PubMed Abstract:
TWEAK (TNF homologue with weak apoptosis-inducing activity) and Fn14 (fibroblast growth factor-inducible protein 14) are members of the tumor necrosis factor (TNF) ligand and receptor super-families. Having observed that Xenopus Fn14 cross-reacts with human TWEAK, despite its relatively low sequence homology to human Fn14, we examined the conservation in tertiary fold and binding interfaces between the two species. Our results, combining NMR solution structure determination, binding assays, extensive site-directed mutagenesis and molecular modeling, reveal that, in addition to the known and previously characterized β-hairpin motif, the helix-loop-helix motif makes an essential contribution to the receptor/ligand binding interface. We further discuss the insight provided by the structural analyses regarding how the cysteine-rich domains of the TNF receptor super-family may have evolved over time.
Organizational Affiliation:
Biogen Idec Inc., 14 Cambridge Center, MA, USA.