The structure of a putative malate/lactate dehydrogenase from E. coli.
Cuff, M.E., Skarina, T., Edwards, A., Savchenko, A., Cymborowski, M., Minor, W., Joachimiak, A.To be published.
Experimental Data Snapshot
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Malate/L-lactate dehydrogenases | 385 | Escherichia coli K-12 | Mutation(s): 9 Gene Names: ybiC | 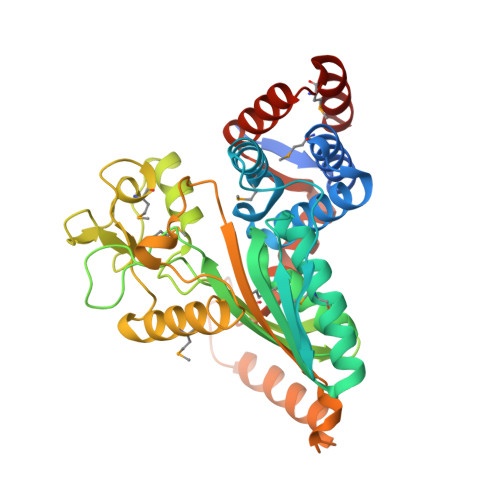 | |
UniProt | |||||
Find proteins for P30178 (Escherichia coli (strain K12)) Explore P30178 Go to UniProtKB: P30178 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P30178 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAD Query on NAD | F [auth A], R [auth B] | NICOTINAMIDE-ADENINE-DINUCLEOTIDE C21 H27 N7 O14 P2 BAWFJGJZGIEFAR-NNYOXOHSSA-N |  | ||
| 1PE Query on 1PE | L [auth A] | PENTAETHYLENE GLYCOL C10 H22 O6 JLFNLZLINWHATN-UHFFFAOYSA-N |  | ||
| SO4 Query on SO4 | C [auth A] D [auth A] E [auth A] M [auth B] N [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| EDO Query on EDO | G [auth A] H [auth A] I [auth A] J [auth A] K [auth A] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| MSE Query on MSE | A, B | L-PEPTIDE LINKING | C5 H11 N O2 Se |  | MET |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 139.095 | α = 90 |
| b = 139.095 | β = 90 |
| c = 151.654 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| SBC-Collect | data collection |
| HKL-2000 | data scaling |
| HKL-3000 | phasing |
| SHELX | phasing |
| MLPHARE | phasing |
| DM | phasing |
| RESOLVE | phasing |
| ARP/wARP | model building |
| O | model building |
| Coot | model building |