The co-crystallisation of (S)-lysine-bound dihydrodipicolinate synthase from E. coli indicates that domain movements are not responsible for (S)-lysine inhibition
Devenish, S.R.A., Dobson, R.C.J., Jameson, G.B., Gerrard, J.A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| dihydrodipicolinate synthase | 292 | Escherichia coli | Mutation(s): 0 EC: 4.2.1.52 | 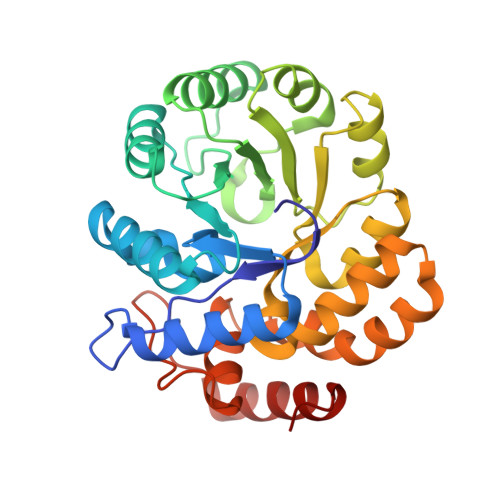 | |
UniProt | |||||
Find proteins for P0A6L2 (Escherichia coli (strain K12)) Explore P0A6L2 Go to UniProtKB: P0A6L2 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0A6L2 | ||||
Sequence AnnotationsExpand | |||||
| |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| DLY Query on DLY | E [auth A], F [auth A], G [auth A] | D-LYSINE C6 H14 N2 O2 KDXKERNSBIXSRK-RXMQYKEDSA-N |  | ||
| K Query on K | C [auth A], H [auth B] | POTASSIUM ION K NPYPAHLBTDXSSS-UHFFFAOYSA-N |  | ||
| CL Query on CL | D [auth A], I [auth B] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 121.68 | α = 90 |
| b = 121.68 | β = 90 |
| c = 109.799 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| AMoRE | phasing |