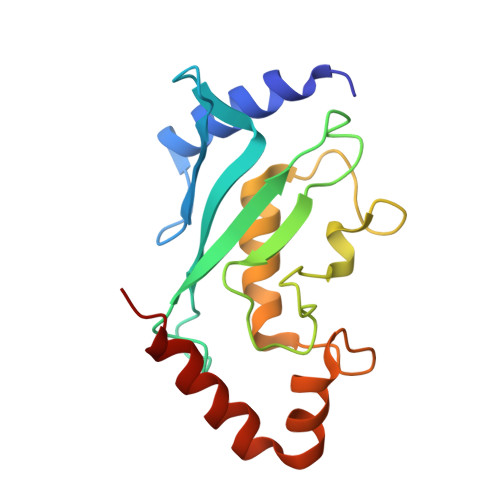
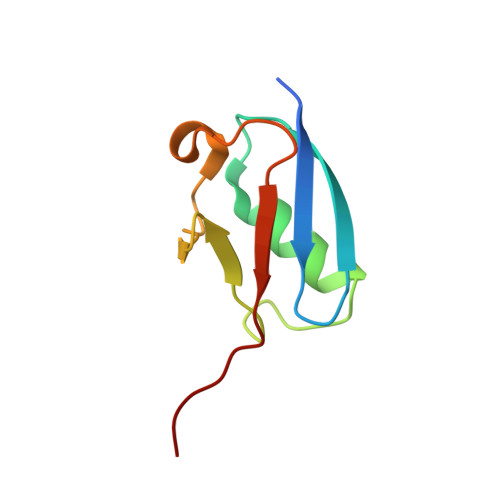
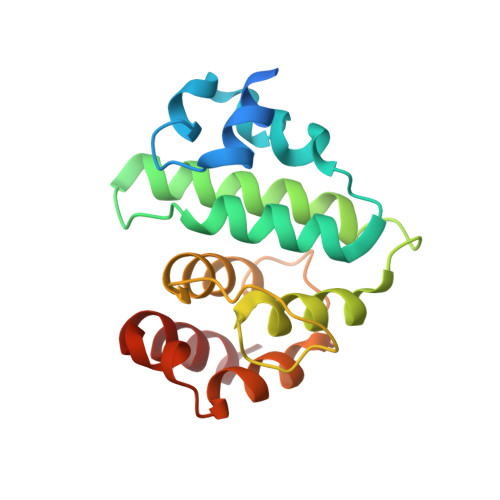
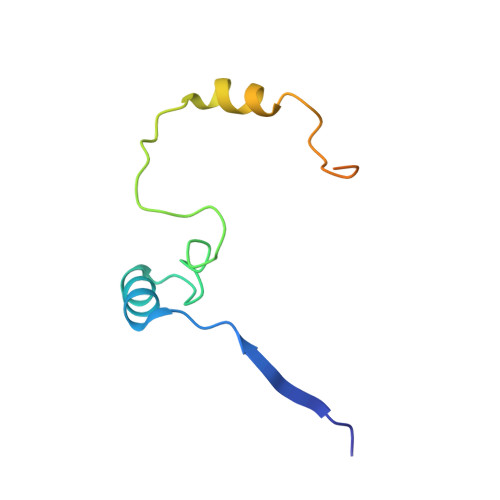
Insights into E3 ligase activity revealed by a SUMO-RanGAP1-Ubc9-Nup358 complex.
Reverter, D., Lima, C.D.(2005) Nature 435: 687-692
- PubMed: 15931224
- DOI: https://doi.org/10.1038/nature03588
- Primary Citation of Related Structures:
1Z5S - PubMed Abstract:
SUMO-1 (for small ubiquitin-related modifier) belongs to the ubiquitin (Ub) and ubiquitin-like (Ubl) protein family. SUMO conjugation occurs on specific lysine residues within protein targets, regulating pathways involved in differentiation, apoptosis, the cell cycle and responses to stress by altering protein function through changes in activity or cellular localization or by protecting substrates from ubiquitination. Ub/Ubl conjugation occurs in sequential steps and requires the concerted action of E2 conjugating proteins and E3 ligases. In addition to being a SUMO E3, the nucleoporin Nup358/RanBP2 localizes SUMO-conjugated RanGAP1 to the cytoplasmic face of the nuclear pore complex by means of interactions in a complex that also includes Ubc9, the SUMO E2 conjugating protein. Here we describe the 3.0-A crystal structure of a four-protein complex of Ubc9, a Nup358/RanBP2 E3 ligase domain (IR1-M) and SUMO-1 conjugated to the carboxy-terminal domain of RanGAP1. Structural insights, combined with biochemical and kinetic data obtained with additional substrates, support a model in which Nup358/RanBP2 acts as an E3 by binding both SUMO and Ubc9 to position the SUMO-E2-thioester in an optimal orientation to enhance conjugation.
Organizational Affiliation:
Structural Biology Program, Sloan-Kettering Institute, New York, New York 10021, USA.