6DZY
Cryo-EM structure of the ts2-active human serotonin transporter in complex with 15B8 Fab and 8B6 ScFv bound to ibogaine
- PDB DOI: https://doi.org/10.2210/pdb6DZY/pdb
- EM Map EMD-8942: EMDB EMDataResource
- Classification: TRANSPORT PROTEIN/IMMUNE SYSTEM
- Organism(s): Homo sapiens, Mus musculus
- Expression System: Homo sapiens, Mus musculus, Escherichia coli
- Mutation(s): No
- Membrane Protein: Yes OPMPDBTMMemProtMDmpstruc
- Deposited: 2018-07-05 Released: 2019-04-24
- Funding Organization(s): Howard Hughes Medical Institute (HHMI), National Institutes of Health/National Institute of Mental Health (NIH/NIMH)
Experimental Data Snapshot
- Method: ELECTRON MICROSCOPY
- Resolution: 4.10 Å
- Aggregation State: PARTICLE
- Reconstruction Method: SINGLE PARTICLE
wwPDB Validation 3D Report Full Report
This is version 2.0 of the entry. See complete history.
Macromolecules
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Sodium-dependent serotonin transporter | 537 | Homo sapiens | Mutation(s): 0 Gene Names: SLC6A4, HTT, SERT Membrane Entity: Yes | 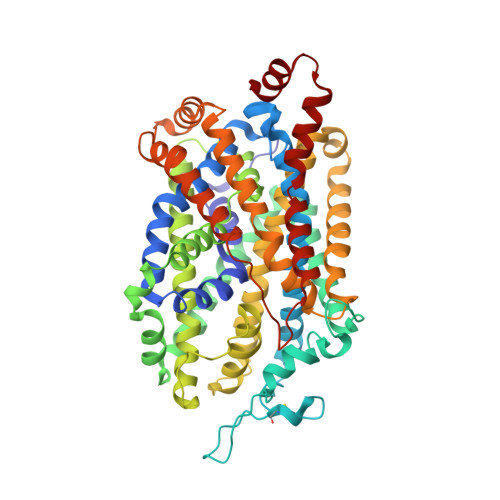 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P31645 (Homo sapiens) Explore P31645 Go to UniProtKB: P31645 | |||||
PHAROS: P31645 GTEx: ENSG00000108576 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P31645 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 15B8 antibody heavy chain | B [auth H] | 118 | Mus musculus | Mutation(s): 0 | 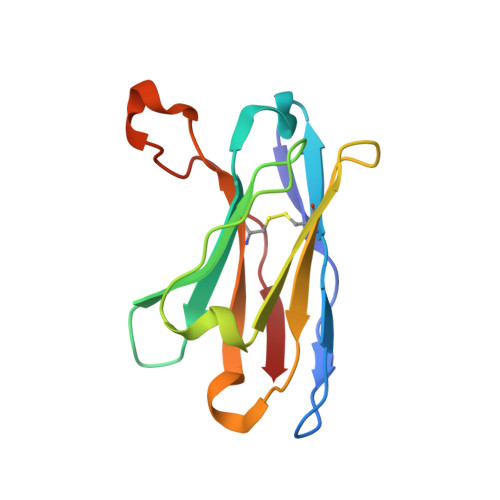 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 15B8 antibody light chain | C [auth L] | 110 | Mus musculus | Mutation(s): 0 | 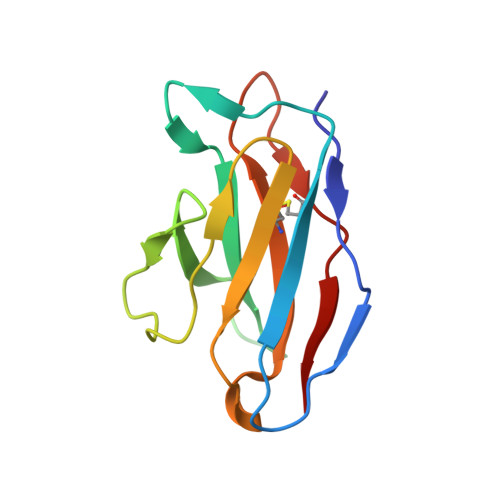 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 8B6 antibody single chain | D [auth V] | 240 | Mus musculus | Mutation(s): 0 | 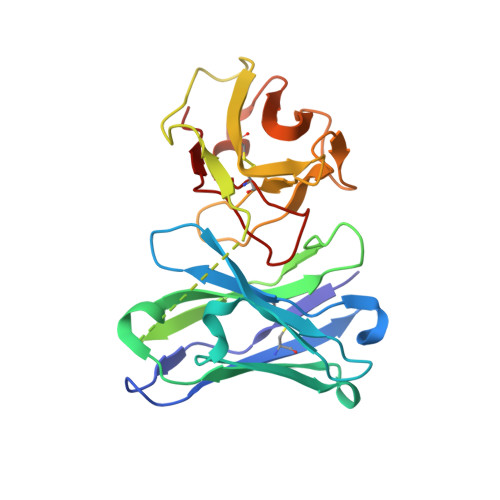 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Oligosaccharides
Small Molecules
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| LMT Query on LMT | G [auth A] | DODECYL-BETA-D-MALTOSIDE C24 H46 O11 NLEBIOOXCVAHBD-QKMCSOCLSA-N |  | ||
| HJM Query on HJM | F [auth A] | (5beta)-12-methoxyibogamine C20 H26 N2 O HSIBGVUMFOSJPD-CFDPKNGZSA-N |  | ||
Experimental Data & Validation
Experimental Data
- Method: ELECTRON MICROSCOPY
- Resolution: 4.10 Å
- Aggregation State: PARTICLE
- Reconstruction Method: SINGLE PARTICLE
Entry History & Funding Information
Deposition Data
- Released Date: 2019-04-24 Deposition Author(s): Coleman, J.A., Yang, D., Gouaux, E.
| Funding Organization | Location | Grant Number |
|---|---|---|
| Howard Hughes Medical Institute (HHMI) | United States | -- |
| National Institutes of Health/National Institute of Mental Health (NIH/NIMH) | United States | 5R37MH070039 |
Revision History (Full details and data files)
- Version 1.0: 2019-04-24
Type: Initial release - Version 1.1: 2019-06-05
Changes: Data collection, Database references - Version 1.2: 2019-11-20
Changes: Author supporting evidence - Version 2.0: 2020-07-29
Type: Remediation
Reason: Carbohydrate remediation
Changes: Advisory, Atomic model, Data collection, Derived calculations, Structure summary














