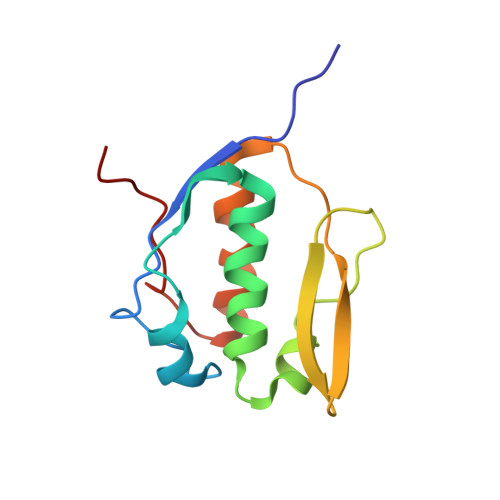
Solution NMR Structure of the DNA-binding Domain from Scml2 (Sex Comb on Midleg-like 2).
Bezsonova, I.(2014) J Biol Chem 289: 15739-15749
- PubMed: 24727478
- DOI: https://doi.org/10.1074/jbc.M113.524009
- Primary Citation of Related Structures:
2MEM - PubMed Abstract:
Scml2 is a member of the Polycomb group of proteins involved in epigenetic gene silencing. Human Scml2 is a part of a multisubunit protein complex, PRC1 (Polycomb repressive complex 1), which is responsible for maintenance of gene repression, prevention of chromatin remodeling, preservation of the "stemness" of the cell, and cell differentiation. Although the majority of PRC1 subunits have been recently characterized, the structure of Scml2 and its role in PRC1-mediated gene silencing remain unknown. In this work a conserved protein domain within human Scml2 has been identified, and its structure was determined by solution NMR spectroscopy. This module was named Scm-like embedded domain, or SLED. Evolutionarily, the SLED domain emerges in the first multicellular organisms, consistent with the role of Scml2 in cell differentiation. Furthermore, it is exclusively found within the Scm-like family of proteins, often accompanied by malignant brain tumor domain (MBT) and sterile α motif (SAM) domains. The domain adopts a novel α/β fold with no structural analogues found in the Protein Data Bank (PDB). The ability of the SLED to bind double-stranded DNA was also examined, and the isolated domain was shown to interact with DNA in a sequence-specific manner. Because PRC1 complexes localize to the promoters of a specific subset of developmental genes in vivo, the SLED domain of Scml2 may provide an important link connecting the PRC1 complexes to their target genes.
Organizational Affiliation:
From the Department of Molecular Biology and Biophysics, University of Connecticut Health Center, Farmington, Connecticut, 06032-3305 bezsonova@uchc.edu.