6CNK
Structure of the 3alpha2beta stiochiometry of the human Alpha4Beta2 nicotinic receptor
- PDB DOI: https://doi.org/10.2210/pdb6CNK/pdb
- EM Map EMD-7536: EMDB EMDataResource
- Classification: TRANSPORT PROTEIN
- Organism(s): Homo sapiens, Mus musculus
- Expression System: Homo sapiens
- Mutation(s): No
- Membrane Protein: Yes PDBTMMemProtMDmpstruc
- Deposited: 2018-03-08 Released: 2018-05-02
- Funding Organization(s): National Institutes of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS), National Institutes of Health/National Institute on Drug Abuse (NIH/NIDA)
Experimental Data Snapshot
- Method: ELECTRON MICROSCOPY
- Resolution: 3.90 Å
- Aggregation State: PARTICLE
- Reconstruction Method: SINGLE PARTICLE
wwPDB Validation 3D Report Full Report
This is version 2.0 of the entry. See complete history.
Macromolecules
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Neuronal acetylcholine receptor subunit alpha-4 | 386 | Homo sapiens | Mutation(s): 0 Gene Names: CHRNA4, NACRA4 Membrane Entity: Yes | 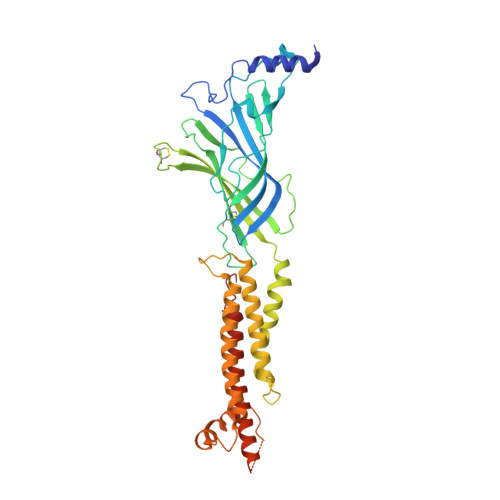 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P43681 (Homo sapiens) Explore P43681 Go to UniProtKB: P43681 | |||||
PHAROS: P43681 GTEx: ENSG00000101204 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P43681 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Neuronal acetylcholine receptor subunit beta-2 | 403 | Homo sapiens | Mutation(s): 0 Gene Names: CHRNB2 Membrane Entity: Yes | 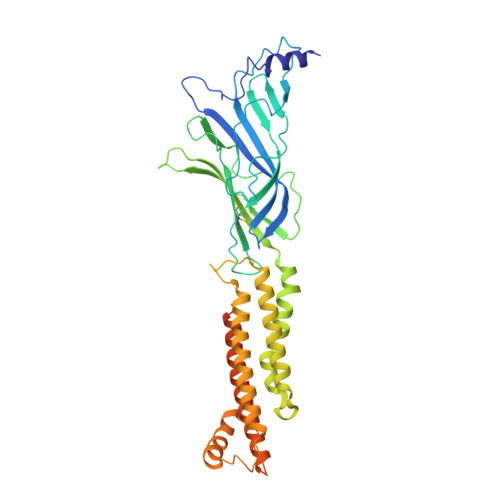 | |
UniProt & NIH Common Fund Data Resources | |||||
Find proteins for P17787 (Homo sapiens) Explore P17787 Go to UniProtKB: P17787 | |||||
PHAROS: P17787 GTEx: ENSG00000160716 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P17787 | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| IgG1 Kappa Light Chain | F, H [auth J] | 238 | Mus musculus | Mutation(s): 0 | 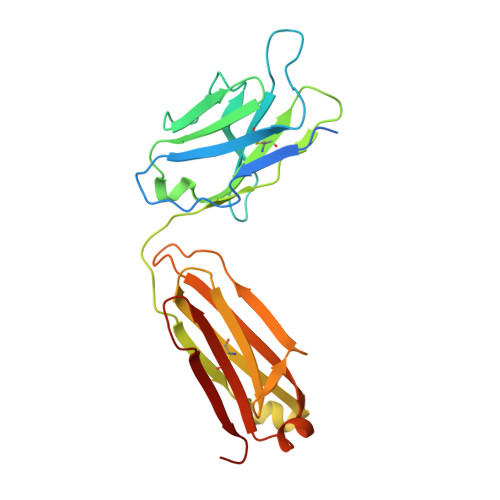 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Find similar proteins by:
(by identity cutoff) | 3D Structure
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| IgG1 Heavy Chain | G, I [auth K] | 462 | Mus musculus | Mutation(s): 0 | 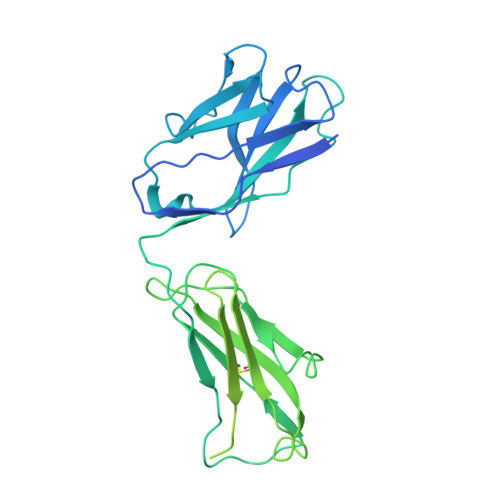 |
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
Sequence AnnotationsExpand | |||||
| |||||
Oligosaccharides
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | 3D Interactions |
| beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | J [auth H], K [auth I] | 3 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G15407YE GlyCosmos: G15407YE GlyGen: G15407YE | |||||
Small Molecules
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| Y01 Query on Y01 | AA [auth E] BA [auth E] M [auth A] N [auth A] S [auth B] | CHOLESTEROL HEMISUCCINATE C31 H50 O4 WLNARFZDISHUGS-MIXBDBMTSA-N |  | ||
| NAG Query on NAG | L [auth A], Q [auth B], X [auth D] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| NCT Query on NCT | P [auth A], R [auth B], W [auth D] | (S)-3-(1-METHYLPYRROLIDIN-2-YL)PYRIDINE C10 H14 N2 SNICXCGAKADSCV-JTQLQIEISA-N |  | ||
| NA Query on NA | O [auth A] | SODIUM ION Na FKNQFGJONOIPTF-UHFFFAOYSA-N |  | ||
Experimental Data & Validation
Experimental Data
- Method: ELECTRON MICROSCOPY
- Resolution: 3.90 Å
- Aggregation State: PARTICLE
- Reconstruction Method: SINGLE PARTICLE
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | RELION | 2.1 |
| MODEL REFINEMENT | PHENIX | 1.13-2988 |
Entry History & Funding Information
Deposition Data
- Released Date: 2018-05-02 Deposition Author(s): Walsh Jr, R.M., Roh, S.H., Gharpure, A., Morales-Perez, C.L., Hibbs, R.E.
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) | United States | NS095899 |
| National Institutes of Health/National Institute on Drug Abuse (NIH/NIDA) | United States | DA037492 |
| National Institutes of Health/National Institute on Drug Abuse (NIH/NIDA) | United States | DA042072 |
Revision History (Full details and data files)
- Version 1.0: 2018-05-02
Type: Initial release - Version 1.1: 2018-05-09
Changes: Data collection, Structure summary - Version 1.2: 2018-05-16
Changes: Data collection, Database references - Version 1.3: 2018-05-30
Changes: Data collection, Database references - Version 1.4: 2018-07-18
Changes: Data collection - Version 1.5: 2019-11-20
Changes: Data collection, Database references - Version 1.6: 2019-12-11
Changes: Author supporting evidence - Version 1.7: 2019-12-18
Changes: Other - Version 2.0: 2020-07-29
Type: Remediation
Reason: Carbohydrate remediation
Changes: Advisory, Atomic model, Data collection, Derived calculations, Structure summary













