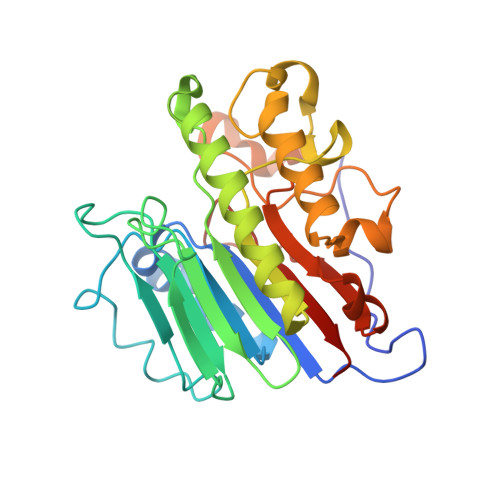
Molecular and structural characterization of disease-associated APE1 polymorphisms.
Whitaker, A.M., Stark, W.J., Flynn, T.S., Freudenthal, B.D.(2020) DNA Repair (Amst) 91-92: 102867-102867
- PubMed: 32454397
- DOI: https://doi.org/10.1016/j.dnarep.2020.102867
- Primary Citation of Related Structures:
6W0Q, 6W2P, 6W3L, 6W3N, 6W3Q, 6W3U, 6W43 - PubMed Abstract:
Under conditions of oxidative stress, reactive oxygen species (ROS) continuously assault the structure of DNA resulting in oxidation and fragmentation of the nucleobases. When the nucleobase structure is altered, its base-pairing properties may also be altered, promoting mutations. Consequently, oxidative DNA damage is a major source of the mutation load that gives rise to numerous human maladies, including cancer. Base excision repair (BER) is the primary pathway tasked with removing and replacing mutagenic DNA base damage. Apurinic/apyrimidinic endonuclease 1 (APE1) is a central enzyme with AP-endonuclease and 3' to 5' exonuclease functions during BER, and therefore is key to maintenance of genome stability. Polymorphisms, or SNPs, in the gene encoding APE1 (APEX1) have been identified among specific human populations and result in variants of APE1 with modified function. These defects in APE1 potentially result in impaired DNA repair capabilities and consequently an increased risk of disease for individuals within these populations. In the present study, we determined the X-ray crystal structures of three prevalent disease-associated APE1 SNPs (D148E, L104R, and R237C). Each APE1 SNP results in unique localized changes in protein structure, including protein dynamics and DNA binding contacts. Combined with comprehensive biochemical characterization, including pre-steady-state kinetic and DNA binding analyses, variant APE1:DNA complex structures with both AP-endonuclease and exonuclease substrates were analyzed to elucidate how these SNPs might perturb the two major repair functions employed by APE1 during BER.
Organizational Affiliation:
Department of Biochemistry and Molecular Biology, University of Kansas Medical Center, Kansas City, KS,66160, USA.