Pyocin S5 Import into Pseudomonas aeruginosa Reveals a Generic Mode of Bacteriocin Transport.
Behrens, H.M., Lowe, E.D., Gault, J., Housden, N.G., Kaminska, R., Weber, T.M., Thompson, C.M.A., Mislin, G.L.A., Schalk, I.J., Walker, D., Robinson, C.V., Kleanthous, C.(2020) mBio 11
- PubMed: 32156826
- DOI: https://doi.org/10.1128/mBio.03230-19
- Primary Citation of Related Structures:
6THK - PubMed Abstract:
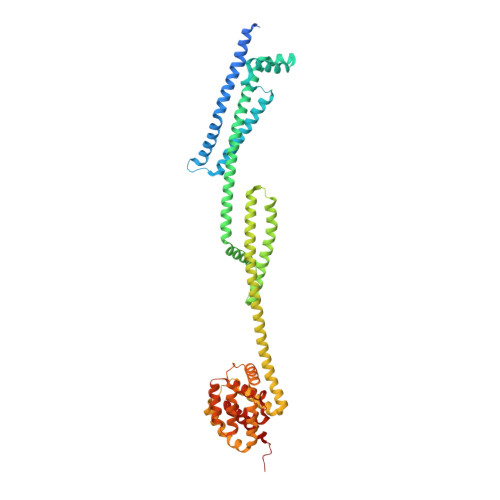
Pyocin S5 (PyoS5) is a potent protein bacteriocin that eradicates the human pathogen Pseudomonas aeruginosa in animal infection models, but its import mechanism is poorly understood. Here, using crystallography, biophysical and biochemical analyses, and live-cell imaging, we define the entry process of PyoS5 and reveal links to the transport mechanisms of other bacteriocins. In addition to its C-terminal pore-forming domain, elongated PyoS5 comprises two novel tandemly repeated kinked 3-helix bundle domains that structure-based alignments identify as key import domains in other pyocins. The central domain binds the lipid-bound common polysaccharide antigen, allowing the pyocin to accumulate on the cell surface. The N-terminal domain binds the ferric pyochelin transporter FptA while its associated disordered region binds the inner membrane protein TonB1, which together drive import of the bacteriocin across the outer membrane. Finally, we identify the minimal requirements for sensitizing Escherichia coli toward PyoS5, as well as other pyocins, and suggest that a generic pathway likely underpins the import of all TonB-dependent bacteriocins across the outer membrane of Gram-negative bacteria. IMPORTANCE Bacteriocins are toxic polypeptides made by bacteria to kill their competitors, making them interesting as potential antibiotics. Here, we reveal unsuspected commonalities in bacteriocin uptake pathways, through molecular and cellular dissection of the import pathway for the pore-forming bacteriocin pyocin S5 (PyoS5), which targets Pseudomonas aeruginosa In addition to its C-terminal pore-forming domain, PyoS5 is composed of two tandemly repeated helical domains that we also identify in other pyocins. Functional analyses demonstrate that they have distinct roles in the import process. One recognizes conserved sugars projected from the surface, while the other recognizes a specific outer membrane siderophore transporter, FptA, in the case of PyoS5. Through engineering of Escherichia coli cells, we show that pyocins can be readily repurposed to kill other species. This suggests basic ground rules for the outer membrane translocation step that likely apply to many bacteriocins targeting Gram-negative bacteria.
Organizational Affiliation:
Department of Biochemistry, University of Oxford, Oxford, United Kingdom.