The U4 Antibody Epitope on Human Papillomavirus 16 Identified by Cryo-electron Microscopy.
Guan, J., Bywaters, S.M., Brendle, S.A., Lee, H., Ashley, R.E., Christensen, N.D., Hafenstein, S.(2015) J Virol 89: 12108-12117
- PubMed: 26401038
- DOI: https://doi.org/10.1128/JVI.02020-15
- Primary Citation of Related Structures:
3JBA - PubMed Abstract:
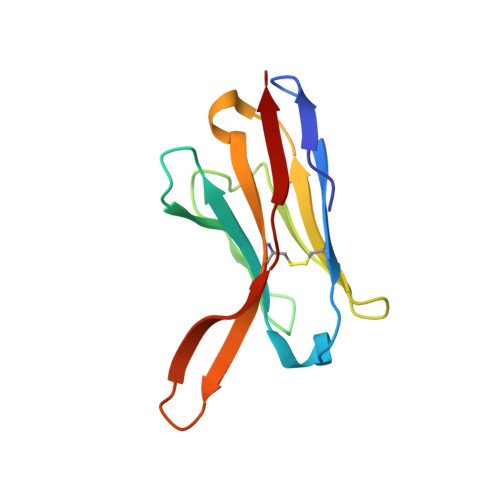
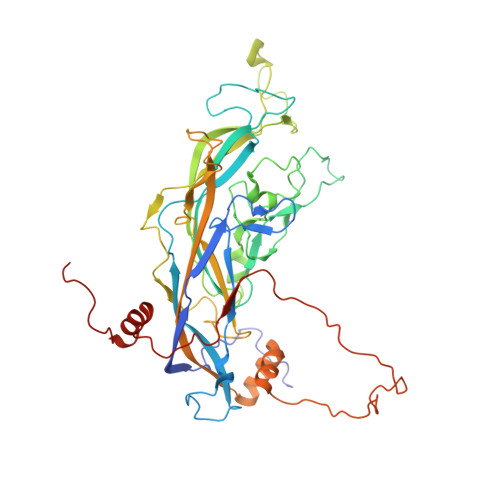
The human papillomavirus (HPV) major structural protein L1 composes capsomers that are linked together through interactions mediated by the L1 C terminus to constitute a T=7 icosahedral capsid. H16.U4 is a type-specific monoclonal antibody recognizing a conformation-dependent neutralizing epitope of HPV thought to include the L1 protein C terminus. The structure of human papillomavirus 16 (HPV16) complexed with H16.U4 fragments of antibody (Fab) was solved by cryo-electron microscopy (cryo-EM) image reconstruction. Atomic structures of virus and Fab were fitted into the corresponding cryo-EM densities to identify the antigenic epitope. The antibody footprint mapped predominately to the L1 C-terminal arm with an additional contact point on the side of the capsomer. This footprint describes an epitope that is presented capsid-wide. However, although the H16.U4 epitope suggests the presence of 360 potential binding sites exposed in the capsid valley between each capsomer, H16.U4 Fab bound only to epitopes located around the icosahedral five-fold vertex of the capsid. Thus, the binding characteristics of H16.U4 defined in this study showed a distinctive selectivity for local conformation-dependent interactions with specific L1 invading arms between five-fold related capsomers.
Organizational Affiliation:
Department of Medicine, The Pennsylvania State University College of Medicine, Hershey, Pennsylvania, USA.